
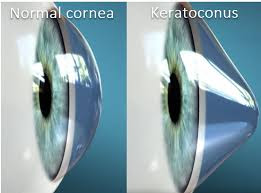
Keratoconus, often referred to as ‘KC’, is a non-inflammatory chronic eye condition in which the typically round dome-shaped cornea progressively thins and weakens, causing the development of a cone-like bulge and optical irregularity of the cornea. This can cause blurred vision and significant visual impairment. Early detection and intervention is key to saving vision with Keratoconus, this is why an annual eye exam is critical for people of all ages. There is no cure for Keratoconus but there are ways to treat it and preserve quality of vision for patients with the disease.
Williamson Eye Center is pleased to offer a specialized Keratoconus Treatment Center in Baton Rouge, Louisiana including FDA approved Corneal Cross-Linking to treat the progression of Keratoconus.
Keratoconus typically first appears in individuals who are in their late teens or early twenties, and may progress for 10-20 years. Each eye may be affected differently but it does generally affect both eyes.
In the early stages of Keratoconus, people may experience:
The cornea is responsible for focusing most of the light that comes into the eye. Therefore, abnormalities of the cornea, such as Keratoconus, can have a major impact on how an individual sees the world, making simple tasks such as driving a car or reading a book very difficult. Keratoconus may result in significant vision loss and may lead to corneal transplant in severe cases.
Glasses and contact lenses may be used to correct vision for patients with Keratoconus. Due to the irregular shape of the cornea, specialty contact lenses such as RGP, Scleral, or Hybrid lenses are recommended. Our doctors at Williamson Eye Center are of the most highly skilled specialty lens physicians in the area and we are proud to be the home of the state of Louisiana's only Fellow of the Scleral Lens Society, Dr. Joshua Davidson.

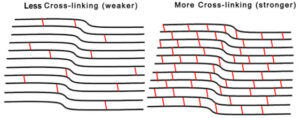
Corneal Cross-linking is a minimally invasive outpatient procedure that combines the use of UVA light and riboflavin eye drops to add stiffness to corneas which have been weakened by disease. The goal of the procedure is to stiffen the cornea to slow or prevent further progression of the condition and preserve vision. Williamson Eye Center is pleased to bring this FDA approved advanced treatment option to patients in South Louisiana.

In severe cases, some patients may require a corneal transplant due to the progression of Keratoconus. A corneal transplant is a surgical procedure to replace part of the cornea with corneal tissue from a donor. In most cases, a corneal transplant is a successful procedure but it does carry a small risk of complications much like any transplant, such as rejection of the donor cornea. Early intervention with Keratoconus is essential. By detecting Keratocoous in an earlier stage and utilizing innovative treatments such as cross-linking early, patients may avoid reaching this stage of severity which could require a transplant.
Glasses:
In the mildest form of Keratoconus, eyeglasses or soft contact lenses may help. But as the disease progresses and the cornea thins and becomes increasingly more irregular in shape, glasses and regular soft contact lenses will no longer provide adequate vision correction.
Specialty Contact Lenses:
As Keratoconus causes a highly irregular shaped corneal surface, normal “soft” contact lenses have a low likelihood of success. Today there are specialized contact lenses, namely “Scleral lenses” that are large-diameter gas permeable contact lenses which are specifically designed to vault over the entire irregular corneal surface while resting gently on the “white” of the eye known as the sclera. This vaulting mechanism allows scleral lenses to essentially replace the irregular cornea with a perfectly smooth optical surface to correct any visual problems caused by Keratoconus or other corneal abnormalities. Due to their larger size (slightly larger than a typical soft lens),the lenses also allow for a more consistent visual outcome as the lens rarely moves on the ocular surface. A scleral lens also acts as a reservoir of saline and tears which provides extreme comfort and can also act as a treatment for those individuals that suffer from severe dry eyes, common in patients with Keratoconus. These lenses have quickly become the treatment modality of choice for thousands of Keratoconic patients including numerous professional athletes.
Corneal Cross-Linking is now FDA approved and greater than 95% of the commercially insured population has access to this potentially sight-protecting treatment.
What is Corneal Cross-Linking?
Cross-linking is a minimally invasive outpatient procedure that combines the use of UVA light and riboflavin eye drops to add stiffness to corneas which have been weakened by disease or refractive surgery. Cross-linking, which has been performed in Europe since 2003, is considered the standard of care around the world for keratoconus and corneal ectasia following refractive surgery. The main goal of Corneal Cross-Linking is to slow progression of the disease and preserve vision.
Corneal Cross-Linking
Riboflavin
Riboflavin (vitamin B2) is important for body growth, red blood cell production and assists in releasing energy from carbohydrates. Its food sources include dairy products, eggs, green leafy vegetables, lean meats, legumes, and nuts. Breads and cereals are often fortified with riboflavin.
Under the conditions used for corneal collagen cross-linking, riboflavin 5’- phosphate, vitamin B2, functions as a photoenhancer which enables the cross- linking reaction to occur.
Ultra-Violet A (UVA)
UVA is one of the three types of invisible light rays given off by the sun (together with ultra-violet B and ultra-violet C) and is the weakest of the three.
A UV light source is applied to irradiate the cornea after it has been soaked in the photo-enhancing riboflavin solution. This cross-linking process stiffens the cornea by increasing the number of molecular bonds, or cross-links, in the collagen.
Patients over the age of 14 who have been diagnosed with progressive keratoconus or corneal ectasia following refractive surgery should ask their doctor about corneal cross-linking. Contact us today to schedule an evaluation for Corneal Cross-Linking by calling 225-274-3937 or request an appointment online.
Is Corneal Cross-Linking Covered by Insurance?
Insurance coverage for FDA approved cross-linking with Photrexa® Viscous, Photrexa® and the KXL® System is now widely available. Greater than 95% of the commercially insured population has access to this potentially sight-protecting treatment.
What can I expect during a Corneal Cross-Linking procedure?
Will I need to be out of my contact lenses for this process?
Yes. Typically, patients are asked by their doctor to stop wearing hard contact lenses prior to surgery for a period of several weeks.
Am I awake during the procedure?
Yes, typically you will be awake during the treatment. You may be given a medication to help you relax and numbing anesthetic drops.
How long does the treatment take?
The actual procedure takes about an hour, but you will be at the office for approximately two hours to allow sufficient time for preparation and recovery before you return to the comfort of your own home.
Does it hurt?
There is some discomfort during immediate recovery but usually not during the treatment. Immediately following treatment, a bandage contact lens is placed on the surface of the eye to protect the newly treated area. After the numbing drops wear off, there is some discomfort, often described as a gritty, burning sensation managed with Tylenol and artificial tears.
What results can I expect from corneal cross-linking?
Can anyone tell by my appearance that I have had cross-linking?
No. There is no change in the appearance of your eyes following cross-linking.
Contact us today to schedule an evaluation for Corneal Cross-Linking by calling 225-274-3937 or request an appointment online.